RESEARCH INTEREST: Chemo-immunotherapy resistance in tumors can be developed by multiple mechanisms including the selection of pre-existing drug-resistant tumor cells, un-touched tumor initiating cells, genetic aberrations, and oncogenic signaling associated with altered cancer cell surface receptors what we referred as “NEO-ANTIGENS”. The last six years of my research in B-cell lymphoma have contributed towards mining several of such molecular targets associated with chemo-immunotherapy resistance development. Our current lab focus is to understand the majority of these parameters primarily those associated with tumor initiating cells, microenvironment and NEO-ANTIGENS mediated chemo-immunotherapy resistance development. We believe, chemo-resistant tumors have a unique expression of NEO-ANTIGENS compared to the chemo-sensitive tumors and B-cells from healthy donors. We will apply a high-throughput next-generation multi-omics approach, CRISPR screening, therapeutic drugs screening, and syngeneic animal model of lymphoma to characterize molecular features of B-cell lymphoma subsets (Figure 1). While in search of NEO-ANTIGENS, we will dissect the functions of RNA-binding & cancer cell-surface specific protein nucleolin in architecting tumor microenvironment through epigenetically regulation of immune-checkpoint proteins (PDL1, CD47, CTLA4). Nucleolin is a nucleolus-specific protein in normal cells; its unique expression on cancer cell surface makes it important as it can be selectively targetable.
Tumor initiating cells represents less than 1% of the total tumor population, have stem cell-like characteristics (elevated expression of Nanog, Oct4, KLF4, Sox2, ALDH1, and ABCC transporters) when compared to rest the 99% of the non-initiating tumor cells. Importantly, tumor initiating cells are resistant to chemotherapeutic agents and few as 100 of these can develop heterogeneous tumor (lymphoma) in the immunocompromised mouse. Our lab focus is to characterize lymphoma-specific tumor initiating cells particularly non-coding RNA and identify key targets to kill these stem cells population. Knowledge from these studies will direct us to develop new therapeutic tools such as monoclonal antibodies, and DNA-based aptamers against selected oncogenic targets.
In parallel to running projects, lab has focus to setup a drug screening animal platform of patient derived xenografts using immunocompromised NOD/SCID mice for molecularly characterized primary tumors. We will also develop a hematopoietic stem cell transplant animal model using CD45.1 and CD45.2 C57BL/6 congenic strains. This congenic system will valued the effects of novel oncogenes on B-cell development and lymphomagenesis. Importantly, new therapeutic tools such as monoclonal antibodies and DNA-based aptamers for oncogenic targets will be developed.
LAB NUMBER: LSS-002
RESEARCH GROUP:
CURRENT OPENING: Research Fellow & JRF position is available for international funded grant “American Society of Hematology”, USA. Interested candidate can write to principal investigator with full CV, one page of career statement and name of three referees.
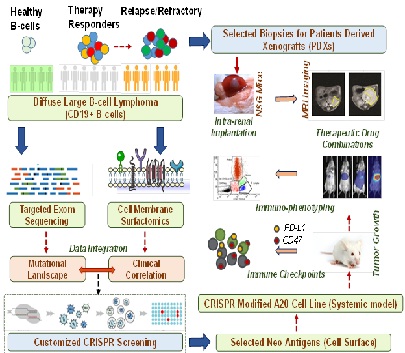
Figure 1: We will identify novel surface receptors “NEO-ANTIGENS” those are specifically present on chemoresistant DLBCL but absent or have low expression in healthy B-cells, or in B-cells derived from chemoresponders. Targeted sequencing will be carried out for the most recurrently mutated genes and epistatic association with NEO-ANTIGENS leading to dismal clinical outcome will be evaluated. We will perform CRISPR screening to screen most critical NEO-ANTIGENS. Using PDX model and syngeneic A20 lymphoma model, we will dissect the impact of NEO-ANTIGENS in modulating tumor microenvironment, and identify novel therapeutic combinations.